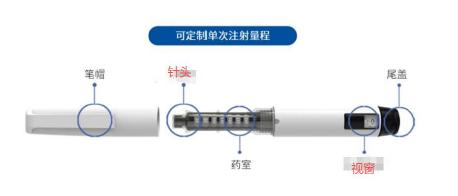
Disposable Auto Prefilled Pen Injector
The Cinvige Disposable Auto Prefilled Pen Injector Assembly is a disposable, easy-to-use, fixed-dose autoinjector with a fully shielded needle. Its core design concept is automatic drug administration. Through a simple three-step operation (unlock, remove, press), it significantly simplifies the injection process and, combined with clear visual and auditory feedback, provides a more reliable and reassuring administration experience for self-injectors, as well as healthcare professionals.

Product Specifications
| Specification | 1.0ml PFS | 2.25ml PFS |
|---|---|---|
| Fill volume | ≤1ml | ≤2.25ml |
| Viscosity | High viscosity / Low viscosity | |
| Type of administration | Fixed-dose | |
| Injection method | Three-step | |
| Feedback system | Two audible clicks (start & end feedback) | |
| Needle insertion depth | 4-7mm | |
| Inject time | ≤15 seconds | |
| Injection force value | ≤15N | |
| Type of needle | 27G, 29G | |
Safety Design
- Full needle protection: The needle remains completely concealed from unlocking through injection completion, preventing accidental needlestick injuries.
- Anti-misoperation mechanism: It adopts a three-step operation process. Injection can only be initiated after unlocking and pressing. This effectively prevents accidental operations
- Structural reliability: Through strict drop tests and vibration tests, it ensures the integrity and reliability of the product during transportation and daily use.
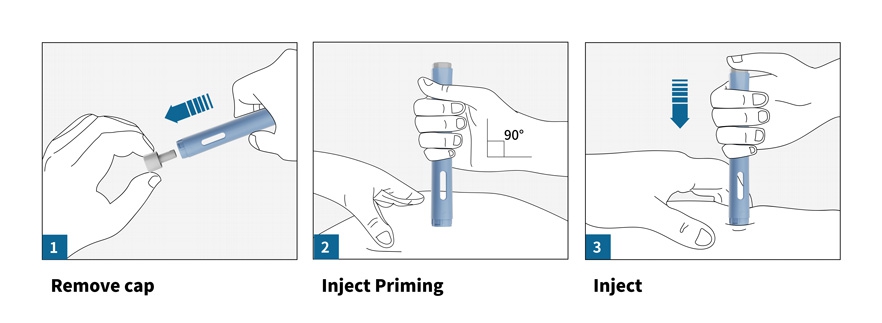
How to Use
Customized Services
We offer flexible and diverse customization solutions to help you create unique products.
- Appearance customization: Supports customization of the color of components such as the syringe observation window, buttons, casing, and pen cap. This takes into account both light-sensitive protection and the need for clinical blinding, enhancing brand recognition.
- Specification Options: Multiple specifications are available to precisely meet diverse clinical and application requirements.
- Technical collaboration: It covers the full range of technical support including process validation, final assembly equipment development, and assembly process optimization, ensuring product performance and client-side production efficiency.
- Platform-based development: Relying on mature core technology platforms to carry out customized development, it significantly shortens the product launch cycle and effectively reduces project R&D risks.
Quality Guarantee
- Compliant with the requirements of the ISO13485 quality management system
- Strict factory inspection standards
- Perfect after-sales service support

Drug Compatibility
Compatible with mainstream drug categories on the market, offering strong adaptability and wide coverage (such as the representative examples below).
GLP-1 receptor agonists
- Dulaglutide
- Polyethylene glycol lorcaserin
- Tilporopeptide
PCSK9 inhibitors (cholesterol-lowering drugs)
- Alirocumab
- Evolocumab
Biological agents / Therapeutic drugs for autoimmune diseases
- Ixekizumab
- Adalimumab
- Erenumab
- Fremanezumab
- Galcanezumab
- Abatacept
- Interferon beta-1a
- Etanercept